 Determine their position on the parent hydrocarbon established in Rule 4. Name alkyl groups, halides and other substituents. Locant - letter or numeral indicating the position of a substituent. 
Part II will cover nomenclature of alcohols, ethers, ketones, aldehydes, carboxylic acids, esters, amides, and amines.įunctional Group - an atom or group of atoms (e.g., a hydroxyl group (-OH)) that replaces hydrogen in an organic compound, defining the structure of a family of compounds and determining properties of the family. This article is written to briefly summarize fundamentals of systematic organic nomenclature with appropriate examples and deals with naming of aliphatic hydrocarbons, haloalkanes and aromatic compounds. As more and more post-secondary institutions teach organic chemistry in first-year courses, the importance of using appropriate nomenclature is clearly apparent. School textbooks are often inconsistent and dated in their application of International Union of Pure and Applied Chemistry (IUPAC) nomenclature rules. Give the parent the same name as if it were an alkane, but replace "_e" with the suffix characteristic of the highest Name the parent hydrocarbon and the principal group(s). 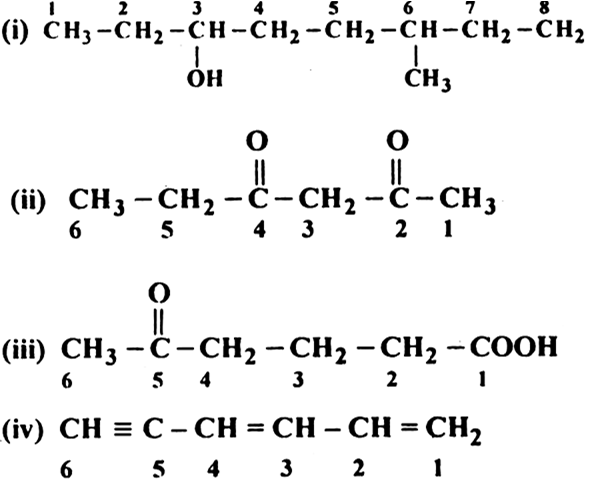
Naming organic compounds is an important component of the Canadian high school chemistry curriculum.' However, highģ. Determine the parent hydrocarbon (principal chain or parent ring system containing the principal functional group). University o(Toronto Toronto ON M5S 3H6 IntroductionĢ. 
Nomenclature of organic compounds - Part I Stanislaw Skonieczny and Andrew P. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
